Three-Dimensional Human Liver Organoids Help Reveal Molecular Secrets of Deadly Ailment
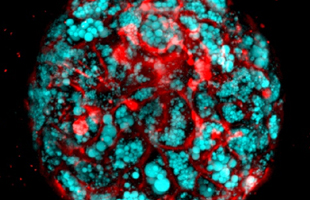 This confocal microscopic image shows a tiny bioengineered human liver organoid grown in the lab with pluripotent stem cells. The mini-organ is accumulating inflammation and fat lipids (bluish bubble-like shapes), which are hallmarks of fatty liver and Wolman disease. |
Scientists at Cincinnati Children’s bioengineered human liver organoids that faithfully mimic key features of Wolman disease in the laboratory setting. This allowed them to uncover underlying disease biology in the organoids and test a potential therapy that in preclinical lab tests reversed the often-fatal childhood condition.
In findings published online by the journal Cell Metabolism (May 30, 2019), researchers say their study overcomes major hurdles to unraveling the molecular mysteries of liver diseases and finding new therapies. The study was a collaboration of scientists at Cincinnati Children’s and five other institutions in Japan and the United States.
“We developed a reproducible method to bioengineer complex, multicellular human liver organoids using pluripotent stem cells derived from healthy human donors and those with liver disease,” said Takanori Takebe, MD, lead study investigator and a physician at the Cincinnati Children’s Division of Gastroenterology, Hepatology and Nutrition and the hospital’s Center for Stem Cell and Organoid Medicine (CuSTOM). “These organoids generate different types of liver cells like hepatocytes and stellate and Kupffer-like cells, and they accurately recreate inflammation, fibrosis and other features of liver disease.”
"We developed a reproducible method to bioengineer complex, multicellular human liver organoids…(that) accurately recreate inflammation, fibrosis and other features of liver disease.”
Takanori Takebe, MD Lead study investigator
That is significant because years of attempted drug development for conditions such as fatty liver disease (NASH, NAFLD) and Wolman disease have been hindered by a lack of human disease models that accurately mimic the diseases, Takebe said. And although certain animal models such as mice are available, they aren’t always reliable for studying human disease.
Finding Possible Solutions
In this study, Takebe and his colleagues used 11 different lines of healthy and diseased pluripotent stem cells to generate the human liver organoids. The diseased pluripotent cells included those grown from donors with a deficiency in the lysosomal acid lipase (LAL) protein. Wolman disease is a type of LAL deficiency.
The researchers treated the organoids with free fatty acids, which helped drive liver fat accumulation, inflammation and fibrosis. The organoids were able to recreate liver disease in the lab in a step-by-step, successive manner similar to Wolman disease in children.
After confirming that Wolman disease in the human organoids was caused by LAL deficiency, the researchers reversed that deficiency by exposing the organoids to a recombinant LAL protein. Next, they leveraged earlier research that shows LAL deficiency causes over-activation of a protein called mammalian target of rapamycin (mTOR), which is linked to liver fibrosis.
Previous research shows that mTOR can be suppressed by a receptor protein called farnesoid X receptor (FXR), so Takebe and his colleagues decided to treat the diseased organoids with an FXR-targeted compound called FGF19. This effectively suppressed fat accumulation in the human liver organoids and prolonged their survival.
The study opens the possibility of further preclinical testing to see whether FXR treatment might be an appropriate strategy for treating Wolman disease, Takebe says. It also could lead to personalized methods to study inflammation and fibrosis in liver disease that match the unique genetics and biology of individual patients.

