Tackling the Treatment of Large Osteochondral Injuries
Early results of a tissue-engineered solution are promising, as in vivo studies in a large animal model are set to be completed soon.
Osteochondral injuries affect about 900,000 patients in the United States each year. “They’re quite common, but treating them is challenging,” says pediatric orthopaedic surgeon Patrick Whitlock, MD, PhD. “Finding a better option for treatment of these injuries is important, because they often lead to early osteoarthritis and significant disability in patients.”
Treatment of large pediatric and adolescent osteochondral injuries remains a major clinical challenge, in part because cartilage doesn’t heal itself easily. Work on a tissue-engineered approach to treating these injuries has continued in Whitlock’s Musculoskeletal Regenerative Medicine Research Laboratory over the past year with funding from the 2017 Angela S.M. Kuo Award he received from the Pediatric Orthopaedic Society of North America.
This team, including James Lin, PhD, Stacey Gruber, MS, Paulomi Ghosh, PhD, and Sumit Murab, PhD, first developed a novel type of artificial scaffolding that would dispense growth factors and proteins to help cartilage regrow. “Our work on this approach to treatment has advanced quickly over the last year,” explains Whitlock. “It’s based on our belief that recapitulating normal developmental mechanisms in a sustainable, tissue-engineered construct may lead to better functional and clinical outcomes, particularly in the adolescent and young adult populations.”
The results thus far provide evidence that serve as a proof of concept for the approach and were highlighted in the journal Biofabrication in February 2018.
This past year Whitlock’s team also filed preliminary intellectual property rights on the methods for producing a tissue-inducing scaffold for the treatment of large osteochondral injuries. In addition, they have begun in vivo studies in a large animal model of the effectiveness of their approach in the treatment of large osteochondral injuries in comparison to traditional treatments.
“We’re looking forward to the results of the in vivo studies,” Whitlock says. “We have high hopes that these findings will help us continue on the path toward a better solution to large osteochondral defects that we may translate clinically to our young patients.”
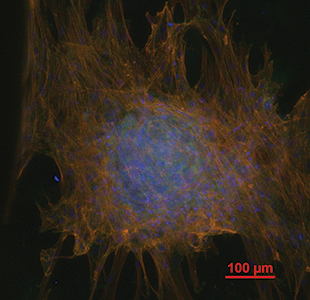
Image description: After 21 days, human mesenchymal stem cells cultured on 3D printed scaffolds containing decellularized cartilage matrix demonstrated mesechymal condensation, a phenomena indicative of chondrogenic differentiation. TRITC-conjugated Phalloidin (red) = actin filaments; DAPI (blue) = cell nuclei; anti-Vinculin monocolonal antibody and FITC secondary antibody (green) = focal contact points.

